POLE3-POLE4 Is a Histone H3-H4 Chaperone that Maintains Chromatin Integrity during DNA Replication - ScienceDirect

Histones H3 and H4 require their relevant amino-tails for efficient nuclear import and replication-coupled chromatin assembly in vivo | Scientific Reports

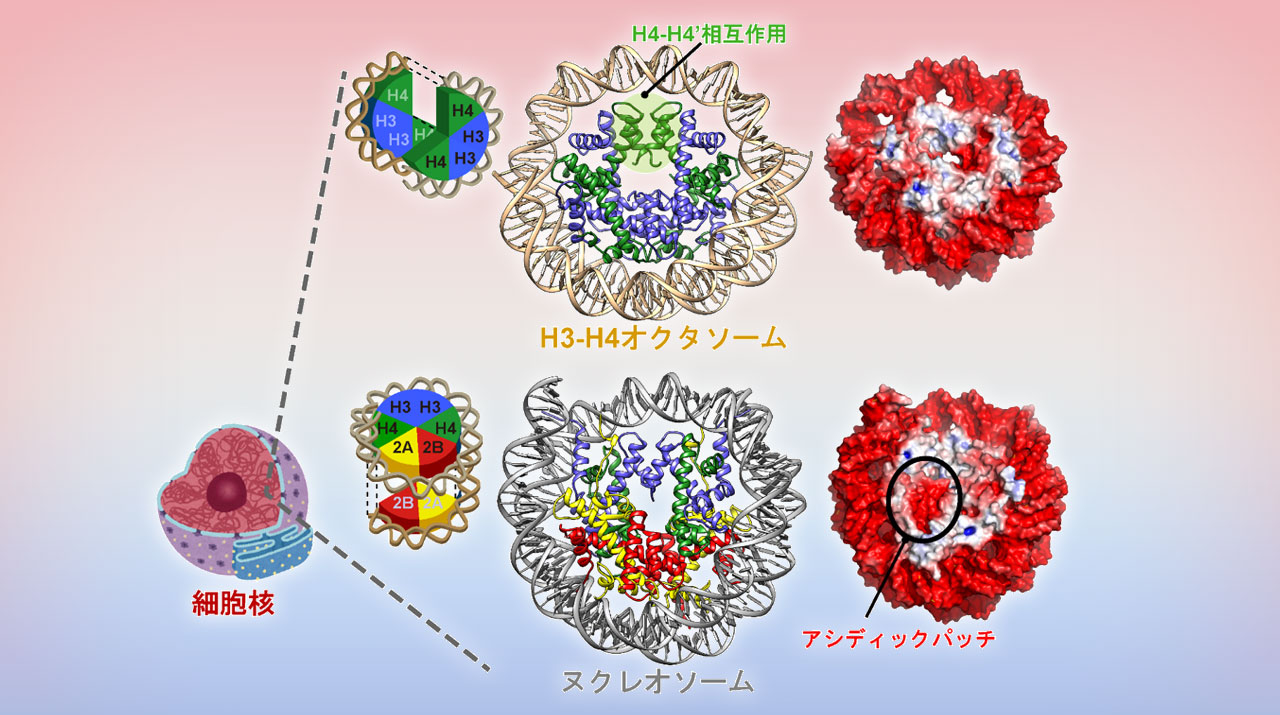
Cryo–electron microscopy structure of the H3-H4 octasome: A nucleosome-like particle without histones H2A and H2B | PNAS

The Histone Chaperones Nap1 and Vps75 Bind Histones H3 and H4 in a Tetrameric Conformation - ScienceDirect

Simultaneous Proteoform Analysis of Histones H3 and H4 with a Simplified Middle-Down Proteomics Method | Analytical Chemistry

Structurally similar motifs in the (H2A-H2B) and the (H3-H4) histone... | Download Scientific Diagram
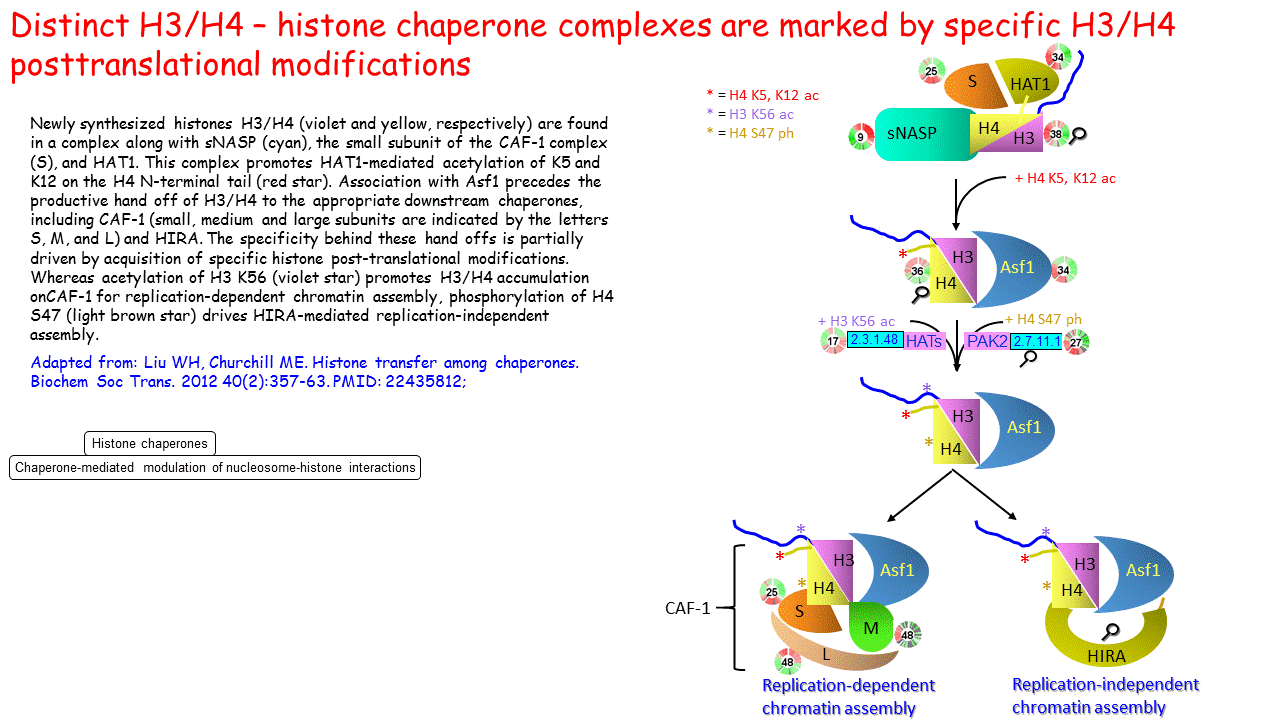
Distinct H3/H4 – histone chaperone complexes are marked by specific H3/H4 posttranslational modifications

Cryo–electron microscopy structure of the H3-H4 octasome: A nucleosome-like particle without histones H2A and H2B | PNAS

Schematic drawing of a nucleosome with the four canonical histones (H3,... | Download Scientific Diagram

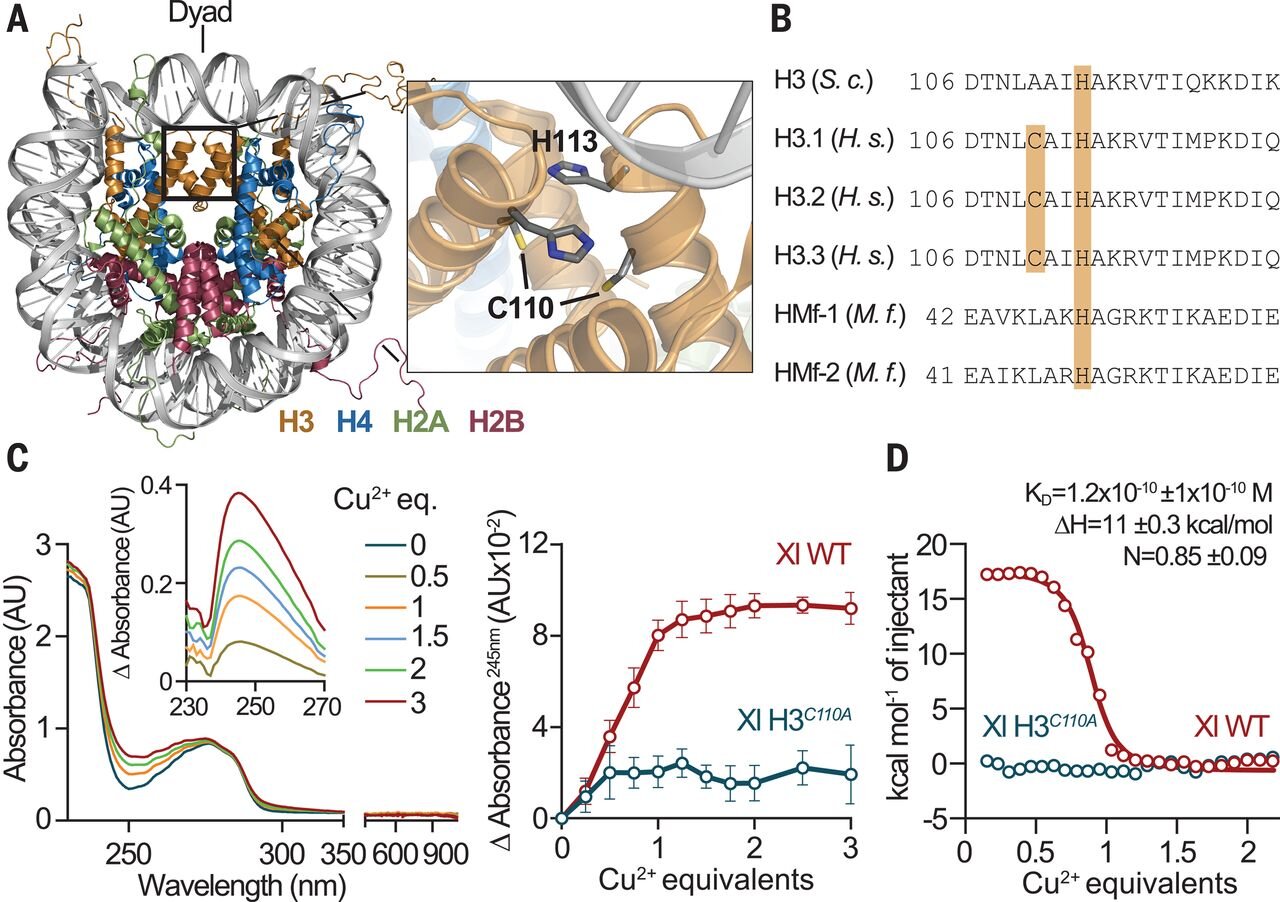
A unique binding mode enables MCM2 to chaperone histones H3–H4 at replication forks | Nature Structural & Molecular Biology